Retinyl Palmitate (Vitamin A); Retinol Palmitate(in 5,804 products)
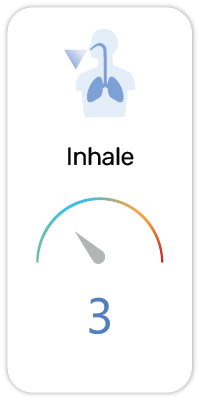
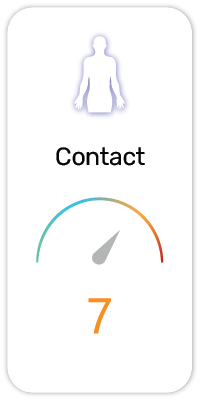
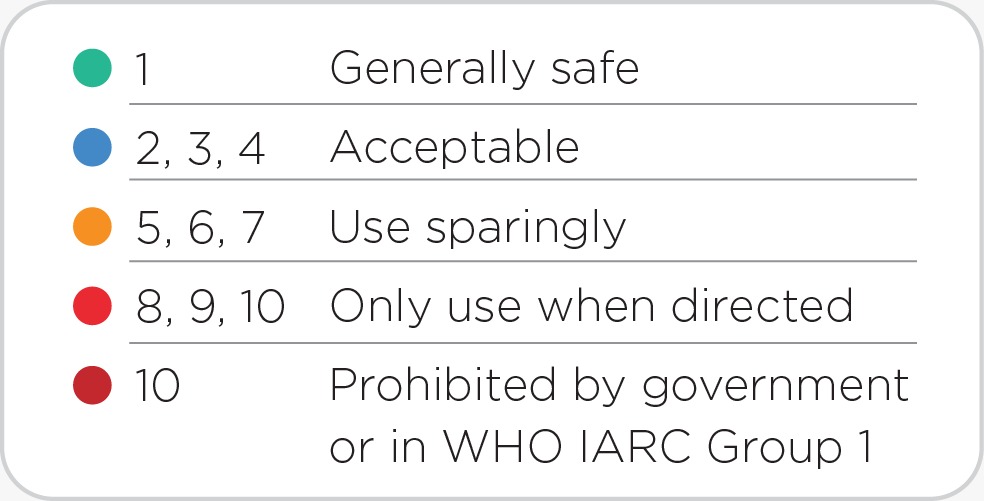
Potential Risk Index®:
About:
Functions:
1. Dietary / Nutritional Supplement - Vitamins, minerals, proteins, fatty acids or probiotics that improves nutritional intake
2. Exfoliant - Removes dead cells at the surface of the skin
Retinyl Palmitate is a naturally-occurring phenyl analogue of retinol (vitamin A) with potential antineoplastic and chemopreventive activities. As the most common form of vitamin A taken for dietary supplementation, retinyl palmitate binds to and activates retinoid receptors, thereby inducing cell differentiation and decreasing cell proliferation. This agent also inhibits carcinogen-induced neoplastic transformation, induces apoptosis in some cancer cell types, and exhibits immunomodulatory properties. [1]
Recent Findings:
Retinyl Palmitate (Retinol Palmitate, RP) is an ester derived from retinol (vitamin A) and palmitic acid. It is commonly used as a dietary vitamin A supplement.
Photodecomposition (the breakdown of a substance by sunlight) of RP is possible via UVA light. [2] [3] DNA fragmentation and cell death were observed "for retinoid concentrations of 100 mM or higher", and when treated
with 150 mM of RP, cell death was observed at 52% of the total. [3] A In another study, RP is classed as a photosensitizer that can be photoactivated by both UVA and UVB light which could "generate short-lived reactive oxygen species that have been shown to damage DNA and proteins and lead to tumors". [3] [4] [5] It has also been noted that RP can be activated even in the presence of visible light (6.3 J/cm^2). [3] It can be seen that retinyl palmitate can be photoactivated in the presence of UVA, UVB and visible light.
Photodecomposition results in a free radical chain reaction that produces free radical compounds such as lipid peroxide among other peroxides. These peroxides are classed as reactive oxygen metabolites and can cause cellular and molecular damage which can possibly result in carcinogenesis. [6] The presence of peroxides has been used as a cancer indicator in both breast and colorectal cancer. [6] [7] Retinoids have been reported to mimic and inhibit the effects of tumor promoters. [8] It is commonly stressed in papers that retinoids do not possess cancerous properties in the absence of light, but its photoactivation and subsequent photodegradation results in free radical compounds that may be cancerous.
Animal trials include the topical addition of retinyl acetate in mice, where there was no conclusive evidence of the carcinogenicity of retinyl acetate. [4] [9] [10] [11] Topical application of 0.005%, 0.025% and 0.05% retinoic acid with UV light causes the development of cutaneous cancers though starting at 0.05% retinoic acid, the retinoic acid concentration "significantly inhibited the tumor formation in this study". [10] Topical application of 0.001% retinoic acid and UV radiation to Skh mice showed significant tumor growth, starting at week 35 compared to the control. [10] The tumors grew so numerously and rapidly that they begin to merge with adjacent tumors. [10] The concentration of retinyl acetate has no effect on photocarcinogenesis. [4]
Studies have shown RP to be to phototoxic to in vitro human Jurkat T-cells (a type of white blood cell). [3] Failure to protect against the photoreactivity of RP may result in photokeratitis of the cornea which could lead to eventual blindness. [4] Long-term human trials on topically applied retinal products have not been carried out although it is currently designated a "high priority compound" by the US National Toxicology Program. [4]
Scientific References:
2. Photodecomposition of Retinyl Palmitate in Ethanol by UVA Light Formation of Photodecomposition Products, Reactive Oxygen Species, and Lipid Peroxides. (Chem. Res. Toxicol., 18(2), 129–138. doi:10.1021/tx049807l)
3. Photo-induced DNA damage and photocytotoxicity of retinyl palmitate and its photodecomposition products. (Toxicol. Ind. Health, 21(5-6), 167–175. doi:10.1191/0748233705th225oa)
4. Photodecomposition and Phototoxicity of Natural Retinoids. (Int. J. Environ. Res. Public Health, 2(1), 147–155. doi:10.3390/ijerph2005010147)
5. Topical Retinoic Acid Enhances, and a Dark Tan Protects, from Subedemal Solar-Simulated Photocarcinogenesis. (J. Investig. Dermatol., 114(5), 923–927. doi:10.1046/j.1523-1747.2000.00861.x)
6. Lipid peroxidation, free radical production and antioxidant status in breast cancer. (Breast Cancer Res. Treat., 59(2), 163–170. doi:10.1023/a:1006357330486)
7. Lipid peroxidation and antioxidant status in colorectal cancer. (World J. Gastroenterol., 11(3), 403. doi:10.3748/wjg.v11.i3.403)
8. Enhancement of experimental photocarcinogenesis by topical retinoic acid. (Cancer Lett., 7(2-3), 85–90. doi:10.1016/s0304-3835(79)80100-7)
9. Retinoids and Photocarcinogenesis: An End to the Controversy? (Photodermatol., 1987, 4, 88-101 doi: 10.1007/978-3-642-60771-4146)
10. Inhibition of Ultraviolet-induced Carcinogenesis by All-trans Retinoic Acid. (J. Investig. Dermatol., 76(3), 178–180. doi:10.1111/1523-1747.ep12525686)
11. Topical All-trans Retinoic Acid Augments Ultraviolet Radiation-Induced Increases in Activated Melanocyte Numbers in Mice. (J. Investig. Dermatol., 112(3), 271–278. doi:10.1046/j.1523-1747.1999.00510.x)
Regulatory References:
1. South Korea - Ministry of Food and Drug Safety - Prohibited/Restricted Chemicals
- Ref: 1223
2. CANADA INGREDIENT HOTLIST, List of Ingredients that are Restricted for Use in Cosmetic Products [2019]
- Retinol and its esters
3. US FDA Generally Recognized As Safe (GRAS) (21 CFR 182) [2017]
Safety and Hazards (UN GHS):
1. Causes skin irritation (H315)
2. May damage fertility or the unborn child (H360)
3. Suspected of damaging fertility or the unborn child (H361)
4. May cause long lasting harmful effects to aquatic life (H413)
Potential Health Concerns For:
1. Abducens Nerve Diseases (PubMed ID:10979654)
2. Abnormalities, Drug-Induced (PubMed ID:8048045)
3. Cardiovascular Diseases (PubMed ID:16015255)
4. Hypervitaminosis A (PubMed ID:10750659)
Potential Health Benefits For:
1. Carcinoma, Squamous Cell (PubMed ID:15984520)
2. Chromosome Aberrations (PubMed ID:32846153)
3. Head and Neck Neoplasms (PubMed ID:15984520)
4. Leukemia, Promyelocytic, Acute (PubMed ID:2388471)
5. Mesothelioma (PubMed ID:16299098)
6. Otitis Media (PubMed ID:17303254)
7. Xerophthalmia (PubMed ID:10979654)
User Comments:
Submit